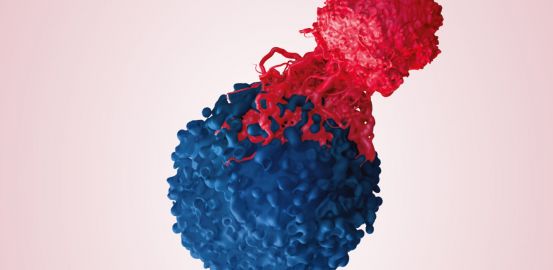
One of the specific features of the ATATIL clinical trial is the production – at CHUV – of lymphocytes that are intended to destroy the tumour. About 15 people work hard in the laboratory to get them to proliferate in time for the infusion.
The white blood cells in a cancer patient’s body survive in a hostile environment. Fighting a particularly aggressive cancer – in vain – they become quite simply exhausted. But once they are brought to the laboratory of CHUV’s Centre for Experimental Therapies, they find themselves in a more favourable environment.
Bathed in interleukin-2, a cell signalling substance that allows their expansion, the lymphocytes recover their strength during an initial phase that may last up to 35 days. After the rejuvenation treatment, they start the second phase of the culture, for 14 days. “We take between 25 and 40 million lymphocytes that are produced during the first phase and again give them preferential treatment,” explains Philippe Gannon, who is in charge of production. The aim, this time, is not only to get them to proliferate, but also to intensely activate the receptors placed on their surface. “The receptor will play a crucial role once the lymphocytes are re-injected into the patient’s body, as they will be at the forefront of intercepting the cancerous cells,” he states.
At the end of these 14 days of careful handling, the “re-boosted” lymphocytes are no longer counted in millions, but in billions: 50 to 75 billion are harvested with extreme caution to be administered to the patient.
“When you observe the technicians’ work, it looks easy, but it’s important to bear in mind that there are years of research behind it all. It’s a real feat to keep cells alive outside the body for such a long time,” emphasises Lionel Trueb, one of the doctors who is looking after the patients included in the trial. All these processes take place in a “clean room”. Although it is not completely sterile, it must contain as few microbes as possible. Each biomedical analysis technician wears a lab coat, gloves, a mask and a hairnet. “In this environment, where concentration is vital, we don’t talk very much and we avoid any unnecessary movement,” Gannon points out.
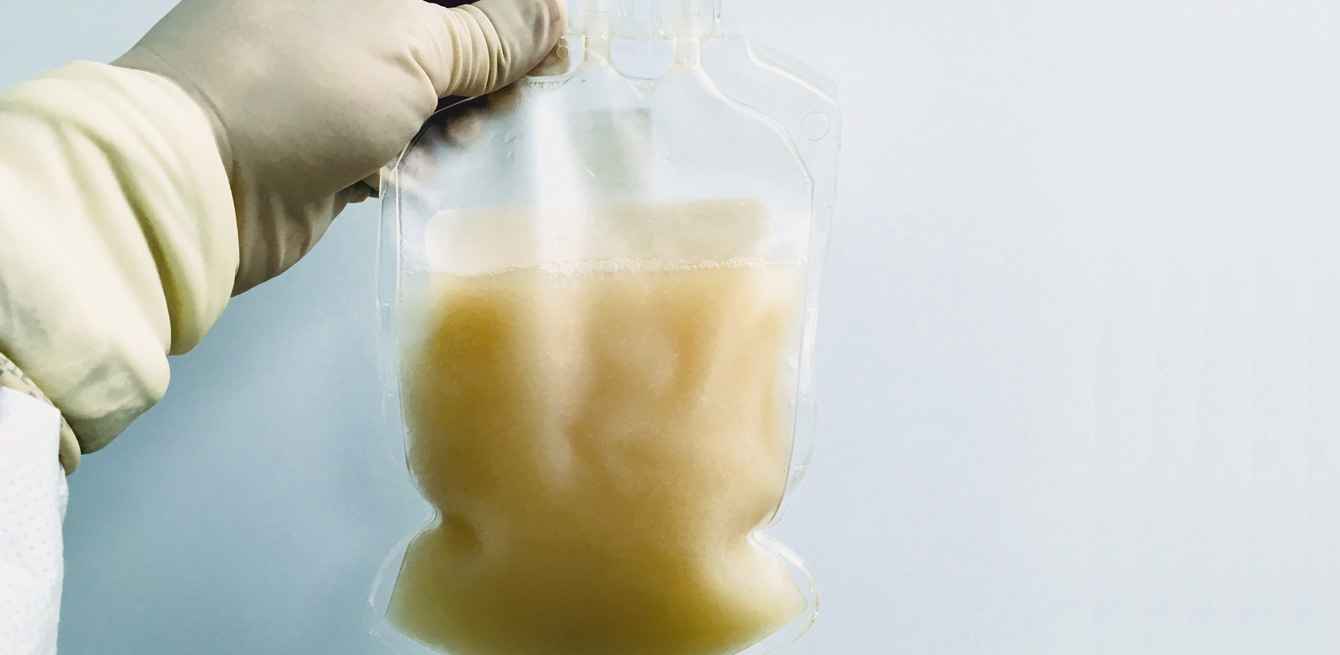
Before they are returned to the patient’s body, the white blood cells still have to be washed and receive a final dose of interleukin-2, so that they are in top condition and remain in a so-called “activated” state. Then comes the quality testing phase. Out of the 300 ml which the infusion bag contains, three are removed to be put through a series of tests, according to a very strict timetable. On the day of re-infusion, the quality control team from the laboratory takes over between 9.30 a.m. and 12 noon. In particular, the team verifies the absence of endotoxins, checks the phenotype and counts the number of cells. Afterwards, all the documentation and the results of the tests are forwarded to Dr Emanuela Marina Iancu, the quality-control manager, who ensures that the process has been observed and that the product complies with the Swissmedic standards. Once she gives the go-ahead, the bag can leave for “reinfusion”.
This is where the lab team’s work ends, but they follow the patients’ progress with interest. “The working conditions are difficult in the lab and the teams like to see tangible results,” explains Gannon. “When I return from symposia on monitoring patients and I announce a reduction in the metastases, there is an amazing energy,” he concludes.