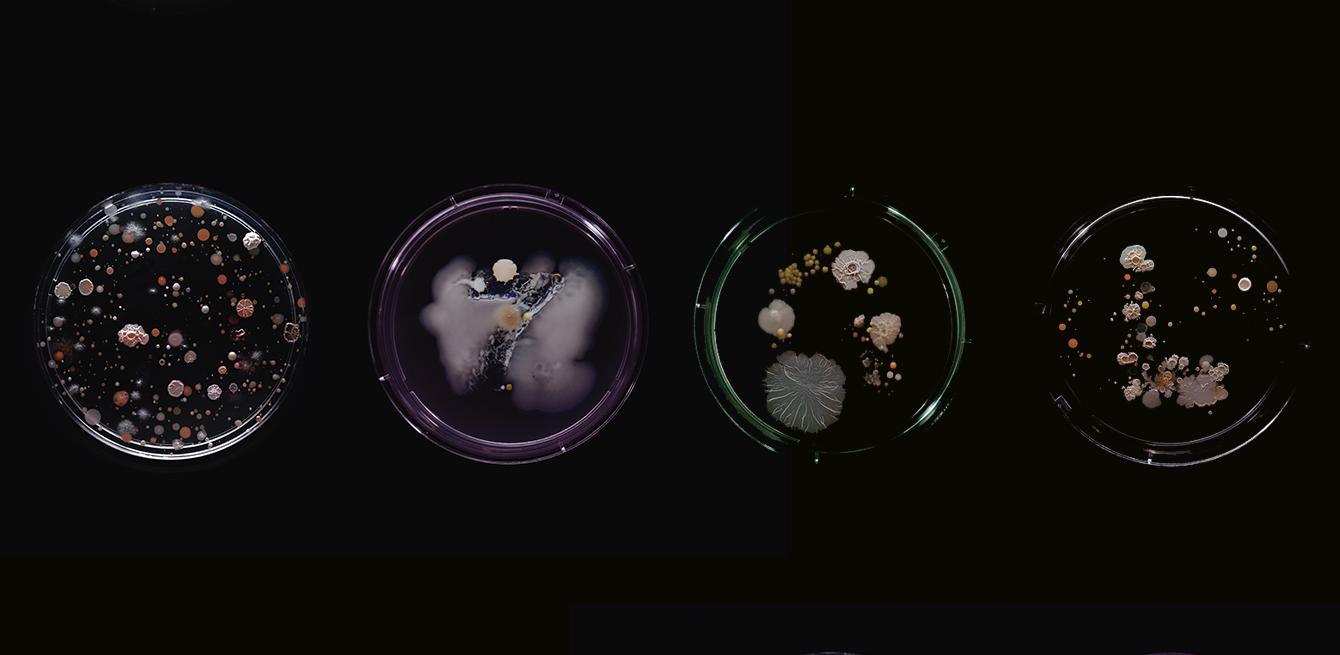
The human body contains 2 kilograms of bacteria. While most are beneficial to health, some cause serious infections that can lead to sepsis or mutate into antibiotic-resistant “super bacteria”. The question remains—are bacteria our friends or our enemies?
The British artist Craig Ward travelled through New York’s twenty-two subway lines in the summer of 2015 to collect bacterial samples. He produced a series of images from his work, demonstrating the complex ecosystem of a city with millions of inhabitants.
Bacteria helped shaped the world as we know it today. You can find them almost everywhere – on doorknobs, in the depths of the ocean and, naturally, in our bodies. There are billions of bacteria in the human body. They function like a fully-fledged organ and we have almost as many of them as we do cells. While most are essential for our health, not all bacteria are our allies. Some types, called pathogenic bacteria, cause infectious diseases that can range from a simple sore throat to a life-threatening case of septic shock.
For many years, antibiotics were an effective way of combating pathogenic bacteria. However, their overuse has led to the development of multidrug-resistant “super bacteria”. At the start of the year, the World Health Organization (WHO) sounded the alarm by reporting that super bacteria could kill up to 10 million people by 2050, placing it on a par with cancer. Scientists are considering several alternative solutions for addressing this emergency. One of them is phage therapy. Discovered a century ago and then abandoned in favour of antibiotics, this method has once again become promising (see article on phagotherapy).
Starting at birth, bacteria colonise our bodies and build what we call the microbiome. “The composition of the bacteria in our bodies changes quite a bit during our first three years of life. It then stabilises to form the microbiome we see in adults,” says Vladimir Lazarevic from the Genomic Research Laboratory at Geneva University Hospitals (HUG). Later, our microbiome changes again when we become elderly due to a weakened immune system and new dietary habits.
Each individual has their own microbiome, which changes depending on one’s environment and diet. The bacteria in the body are mostly contained in the skin or mucous membranes, i.e. the digestive, respiratory, and uro-genital tracts. They exist in symbiosis with the body, which provides them with nutrients and an ideal place to survive and grow. According to Lazarevic, our intestinal tissue and immune system do not develop normally without bacteria.
“Bacteria serve a metabolic purpose. For example, through a process of fermentation, they break down indigestible food residue, such as fibre, into useful components the body can absorb. They can also produce energy substrates like fatty acids and vitamins K and B12,” says the researcher. Bacteria also stimulate the immune system and make the body more resistant. A pathogenic micro-organism, such as a fungus or bacteria, must first fight against the body’s local flora to colonise the environment. These endogenous bacteria are, so to speak, our allies.
However, there are also bacteria that are pathogenic to the human body. These bacteria can cause disease.
“Out of the 13,000 scientifically recognised bacterial species, around 150 are pathogenic to humans,” says Lazarevic.
The best-known types include Staphylococcus, Streptococcus, Clostridium difficile, and Enterobacteria such as Salmonella and Escherichia coli.
“If these bacteria successfully invade and reproduce, they cause disturbances in the body,” explains Thierry Calandra, head of the Infectious Disease Service at CHUV. “An infectious disease is caused when a pathogenic agent encounters the host organism.” However, the presence of pathogenic bacteria in the body doesn’t always result in a reaction. “For example, some people carry Staphylococci in their nasal passages without getting sick,” says Vladimir Lazarevic.

Infectious diseases can range from small, benign, localised infections, such as a sore throat or a bladder infection, to more serious conditions that can even be life threatening. Sepsis is the most serious level of infection. It consists of a generalised inflammatory response in the patient’s body as a result of a severe infection.
According to the WHO, sepsis is a major cause of maternal and neonatal morbidity and mortality in low-income countries. However, it also affects millions of hospitalised patients in higher-income countries; these areas are seeing a rapid increase in the number of cases as the population ages and the number of immunodeficient patients increases.
The bacteria that are most likely to cause sepsis include Streptococcus, Staphylococcus, Enterobacteria, and Pseudomonas. Sepsis can develop due to any kind of severe systemic infection via a number of ways. It can be the result of a delayed response in the host’s body to an infection that the immune system did not immediately recognise. The infection can then spread to such an extent that the body is no longer able to stop it.
In other cases, “the host can react so violently to the infection that it can harm the body,” says Calandra. These excessive inflammatory reactions can cause organ failure and other serious consequences, including the loss of a limb, kidney or pulmonary damage, loss of motor function and neurological disorders.
Septic shock is the most severe form of sepsis. It causes blood pressure to drop precipitously and stops blood flow to the body’s organs. According to Calandra, 10% of septic patients die from the condition; this figure increases to 40% in the case of septic shock.
Even though immunodeficient patients are the most vulnerable to sepsis, he points out that “being young and healthy doesn’t prevent the immune system from failing. If you’re unlucky enough to come across a bacterium that breaks through your defences, you could become septic.”
The specialist goes on to explain that it’s not always easy to spot the preliminary symptoms of sepsis because they can resemble the flu. “You should immediately contact your doctor or go to the emergency room if you start shivering, develop a high fever, and experience general discomfort and an altered mental state. These symptoms indicate sepsis.
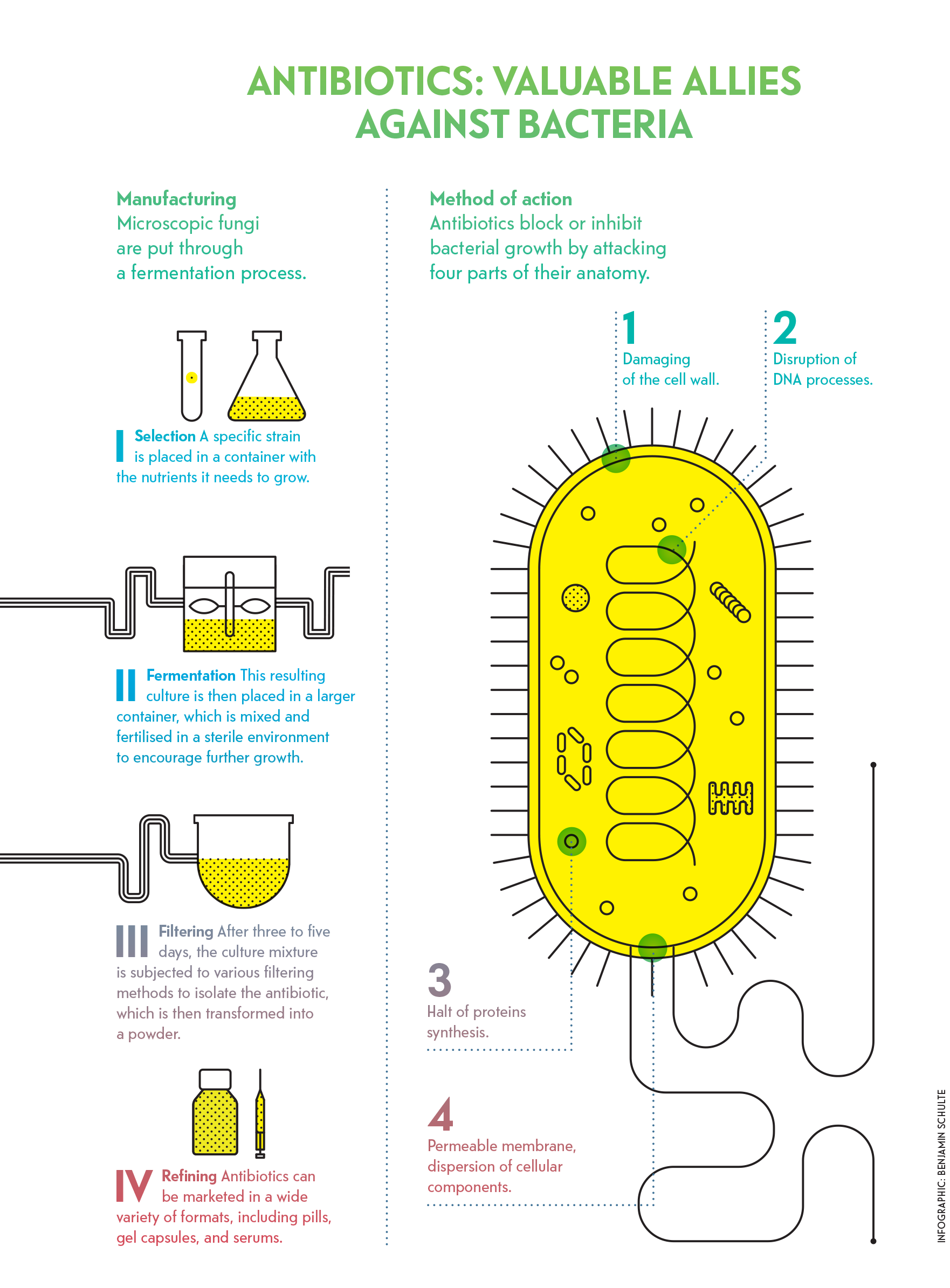
Antibiotics: valuable allies against bacteria

Nowadays, antibiotics are still our most common weapon against bacterial infections. This class of medication became widespread following WWII and represents one of the most significant medical breakthroughs in the twentieth century. “Antibiotics have a spectrum of activity. Some have ‘holes’ in their spectrum, meaning they aren’t effective against all types of bacteria.
A team from the University of Illinois in the United States developed a biochip that can diagnose sepsis using a single drop of blood. Because it often takes some time to diagnose the disease, researchers created a device that can count the number of leucocytes and detect the expression of CD64 on the surface of neutrophils, which are a kind of white blood cell. These two indicators are strong evidence for sepsis. The small device has been tested to verify its reliability and the results were published in “Nature Communications” this summer.
Broad-spectrum antibiotics, however, can stop the growth of a large number of bacteria belonging to a variety of classes,” explains the head of the Infectious Disease Service at CHUV. Antibiotics don’t discriminate between beneficial bacteria and harmful varieties. As a result, antibiotic use can cause side effects. “For example, they can disrupt a person’s intestinal flora and lead to diarrhoea,” says Calandra.
Antibiotics have significantly reduced mortality throughout the world. Without them, certain surgeries such as heart transplants would be impossible. However, their success has also led to an increase in antibiotic resistance over the past few years. According to the WHO, this trend is currently one of the most serious threats to global health. An estimated 700,000 people die every year due to antibiotic-resistant pathogenic agents. At the start of the year, a woman died in the United States due to multidrug-resistant bacteria despite taking over twenty different antibiotics.
Bacteria can develop resistance to an antibiotic through a mutation in its genome or by acquiring resistant genes from another type of bacteria. “Some mutations increase the expression of efflux pumps, i.e. the mechanism that allows cells to evacuate toxic compounds like antibiotics,” says Lazarevic. “Other mutations prevent the antibiotic from entering the cell or change the structure of the medication’s target.” The selective pressure created by the antibiotic leads to the survival and growth of the resistant strain.

When antibiotics are no longer effective, doctors turn to other treatment options. “When the infection is localised, surgery is a possibility,” says Calandra. “For example, if a tuberculosis infection is resistant to all antibiotics, you can remove the part of the lung that’s affected.”
Another alternative is phage therapy, which uses phages, natural viruses that only attack bacteria, to treat infections. “The virus attaches itself to the surface of the bacteria and injects its genetic material inside the cell to reproduce,” says Grégory Resch, project director at the Department of Fundamental Microbiology at the University of Lausanne (UNIL). “The new phages then explode out of the bacteria.”
This treatment was discovered exactly 100 years ago by the French scientist Félix d’Hérelle and was widely used throughout the world prior to the discovery of antibiotics. It is still common in post-Soviet states such as Georgia, Russia and Poland. A specific cocktail of viral phages must be prepared for each species of bacteria.
Bacteriophages are now administered on a much more localised basis to infected injuries such as wounds and burns. They can also be administered through inhalation to treat pulmonary infections or through instillation to treat eye infections. This type of treatment is available over the counter in the aforementioned countries. In Western countries, where this technique was abandoned in favour of antibiotics, clinical trials are currently being conducted to study its validity. ⁄
The number of bacteria present on our skin.
The probability of identifying an individual based on their intestinal microbiome according to a study from the Harvard School of Public Health.
The estimated number of existing bacteria.
This term refers to all micro-organisms (yeast, bacteria and viruses) that are invisible to the naked eye and located within a specific environment (skin, intestine, mouth, etc.).
A microbe is a living organism that can’t be seen with the naked eye. The term includes various life forms, including bacteria, fungi and single-cell algae. Some people also include viruses in this category. However, not everyone in the scientific community considers viruses to be alive.
Bacteria are living, single-cell microscopic organisms that do not have a nucleus. Bacteria were the first life forms to appear on Earth around 3.5 billion years ago.
A virus is a small, infectious, microscopic particle that requires a host, in theory a cell, to reproduce and survive. In this respect, it can be considered a parasite. The debate is still open as to whether or not viruses can be considered living organisms.
Several million children throughout the world die every year from sepsis. In Switzerland, an average of one child per day contracts a potentially deadly infection according to a national, four-year study conducted by the ten largest paediatric hospitals in Switzerland, including CHUV. The results were published on 20 July 2017 in “The Lancet Child & Adolescent Health”, a specialised medical journal. The majority of these infections are contracted at the hospital.
“Following a study conducted by the Swiss Pediatric Sepsis Group, we noted that out of
the 1,181 recorded cases of blood infection, 32% involved previously healthy children, 34% affected newborns and 34% involved children with underlying co-morbidities,” says Sandra Asner, manager of the Paediatric Vaccination and Infectious Disease Unit at CHUV. Asner took part in the study involving pneumococcal disease. “A third of the hospital-acquired infections mostly affect children undergoing chemotherapy, meaning they have a compromised immune system, as well as children in intensive care and premature babies. These three groups are especially at risk of sepsis due to their frequent hospital stays and the use of catheters, which are risk factors for infection.”
Bacteria are typically classified based on two characteristics, namely their shape and the thickness of their cell walls.

is made up of a chain of cocci
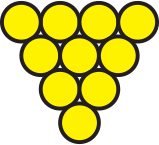
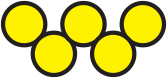
is made up of a cluster of cocci



which causes Lyme disease is transmitted through tick bites

Some self-tanning lotions contain an organic compound made with an industrial bacterial fermentation process.
The work of British artist Anna Dumitriu is entirely sewn using microbial strains, which are sterilised before being integrated into the fabric. For example, one of the artist’s projects involved weaving a strain of Staphylococcus into a blanket.
Some countries, including China, use animal or human excrement to produce domestic biogas through bacterial fermentation.
To measure the thickness of a bacteria’s cell wall, scientists use dyes. Bacteria with thick walls turn blue or purple. These are called Gram-positive bacteria. On the other hand, bacteria with thin walls turn pink or red when dyed. These are called Gram-negative.